Homology modelling and structure-based screening of phenylacetamide derivatives targeting KLK-15 implicated in prostate cancer angiogenesis
DOI:
https://doi.org/10.5530/ctbp.2026.2.16Keywords:
Kallikrein, Angiogenesis, Homology, Virtual screeningAbstract
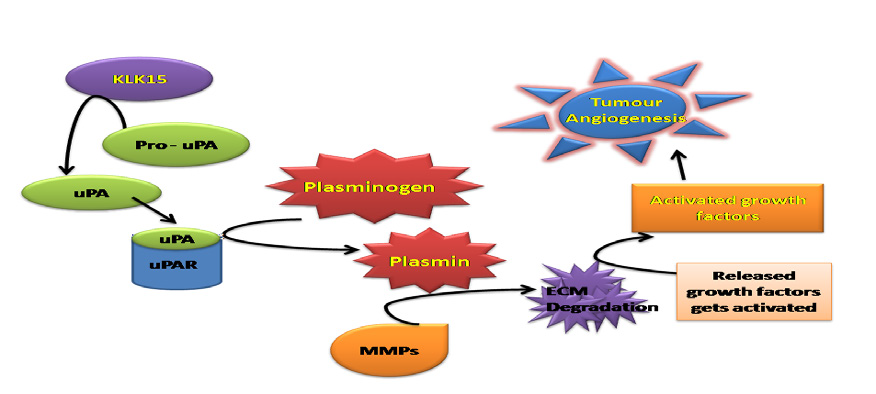
Kallikrein like peptidase-15 (KLK-15) is a human tissue kallikrein protein, found to be expressed in prostate cancer through angiogenesis. KLK-15 acts on pro-uPA which initiates the activation of uPA, uPAR, by converting plasminogen into active plasmin, a proteolytic enzyme. The release of plasmin by KLK-15 is primarily accountable in support of Extracellular Matrix (ECM) degradation, and role of angiogenesis in cancer. Hence, using the KLK-15 protein as a novel target, we focussed on the study of its interaction with phenyl acetamide derivatives as anti-angiogenic agents. The current work involves the generation of 3D model of KLK-15 protein (256 amino acids) by using the homology modelling technique. The 3D model of the KLK-15 protein is MD Simulated and validated using Procheck, ProsA and Verify 3D server tools showing 91.04%, Z-Score = -6.03 and 80.86% parameters indicating the overall reliability of the generated model. Protein-Protein docking with its natural substrate (prouPA) identifies the active site residues. Virtual Screening using phenyl acetamide derivatives was performed by AutoDock 4.2 used through PyRx. The phenyl acetamide derivatives are likely to bind to the KLK-15 protein at His62, Tyr98, Arg101, His148, Glu206, and Ser209 amino acid residues. Based on the binding energy and physico-chemical data ligand L4:N- {5-[(3-methoxy benzyl)sulphanyl]-1,3,4- thiadiazol- 2-yl}-2-2(4-methoxyphemnyl) acetamide, L7: N-{5-[(2-chlorobenzyl)sulfanyl]-1,3,4-thiadiazol- 2-yl}-2-(4-methoxyphenyl) acetamide and ligand L10: N-{5-[(3-chlorobenzyl)sulfanyl]- 1,3,4-thiadiazol-2-yl}-2-(4-methoxyphenyl) acetamide, may act as strong inhibitors against KLK-15 protein and act as a lead molecules against the pathological angiogenesis in prostate cancer.