The Development and Assessment of Senna tora Phytosomal Gel for Dermatophytosis
DOI:
https://doi.org/10.5530/ctbp.2026.2.17Keywords:
Phytosomal gel, Senna tora, Dermatophytosis, Trichophyton rubrum, antifungal activity, skin permeabilityAbstract
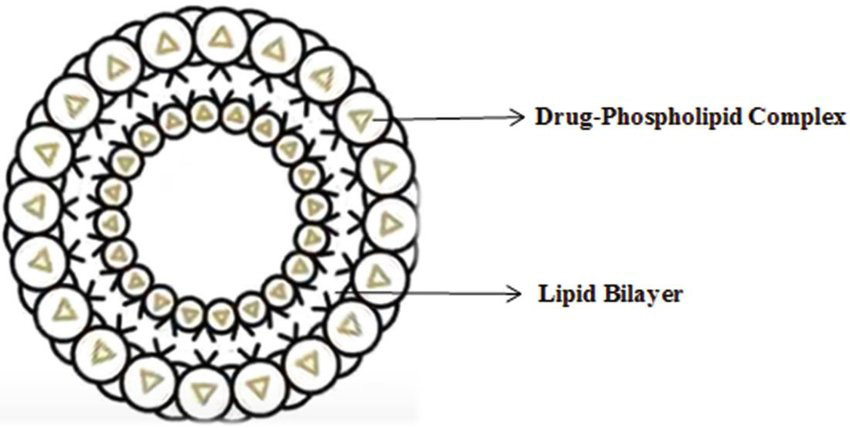
A novel drug delivery system offers an innovative approach to overcoming the limitations of traditional drug formulations. The efficacy of Unani medicine depends on delivering an optimal concentration of therapeutically active compounds. Phytosomes, a vesicular drug delivery system, have been shown to enhance the absorption and bioavailability of herbal extracts, particularly in topical applications. Senna tora (Tukhm-e-Panwar) seeds contain anthraquinone glycosides, primarily Chrysophanic acid-9-anthrone, which exhibit significant antifungal activity. Dermatophytosis (Qooba) is a widespread public health concern, affecting 20-25% of the global population, as per WHO estimates. This study aimed to develop and evaluate a phytosomal gel containing Senna tora seed extract for its antifungal effectiveness in treating Dermatophytosis. Phytosomes were formulated using the anti-solvent precipitation method, with varying Senna tora seed extract-to-soya lecithin ratios. Among the three batches, Batch P1 (1:1 ratio) was optimized based on morphology, percentage yield, entrapment efficiency, and drug content. The optimized Batch P1 was further characterized for zeta potential (-55.3 mV), particle size (196.9 nm), entrapment efficiency (95.44%), and drug content (87.11% w/w). Additionally, Differential Scanning Calorimetry (DSC), Fourier Transform Infrared Spectroscopy (FTIR) for compatibility, Scanning Electron Microscopy (SEM), and in vitro drug diffusion study were conducted. Subsequently, four batches of phytosomal gel were prepared using 0.4% w/w of optimized Batch P1, with varying concentrations (1.25% w/w to 2% w/w) of Carbopol 934 as a gelling agent. These formulations were evaluated for organoleptic properties, homogeneity, pH, drug content, spreadability, extrudability, viscosity, and in vitro release. Batch PG1 was identified as the optimized formulation based on the results and was further assessed for antifungal activity against Trichophyton rubrum and stability. The optimized phytosomal gel Batch PG1 exhibited a visually appealing appearance, a pH of 6.5, a drug content of 96.45%, and an in-vitro drug release of 81.09%. It demonstrated enhanced skin permeability and strong antifungal activity against Trichophyton rubrum, reinforcing its potential as an effective topical treatment for dermatophytosis.