Production and Characterization of Bioactive Peptides by Purified Protease Isolated From Bacillus Sp. Mtcc 9558
DOI:
https://doi.org/10.5530/ctbp.2021.3s.35Keywords:
Bioactive peptides, Antioxidant, Antithrombotic, Tricine-SDS-PAGE, LC-MS-MS analysisAbstract
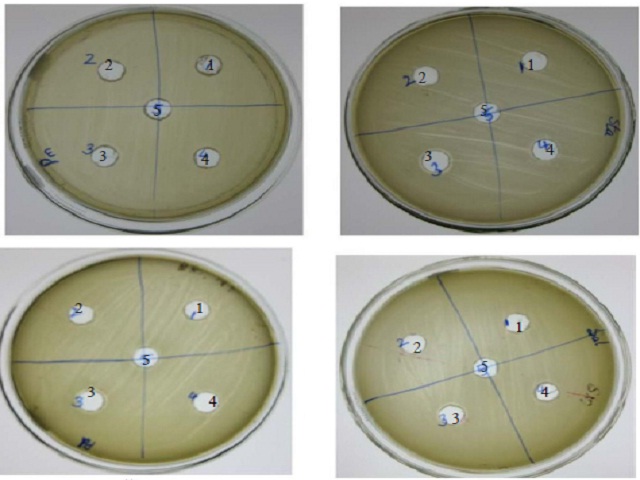
Enzymes are extensively studied for its use in production of bioactive peptides, and due to their great potential they find enomorus commercial applications. The study is use of purified protease in protein hydrolysis for bioactive peptide production and its applications. The protein rich natural sources such as spirulina, yeast, casein, soy protein and β-lactoglobulin were selected and were prepared as protein substrates of protease. Upon reaction, the peptides samples were analysed for their antioxidant, antimicrobial and antithrombotic properties. Antioxidant activities and potential of the protease from Bacillus sp. to generate antibacterial peptides from different natural protein sources was also studied. The results of antithrombotic activity of bioactive peptides from different protein sources indicated inhibitory activities on the thrombin-catalyzed coagulation of fibrinogen. The potent inhibitory effects were observed for casein peptides. The peptides were termed bioactive peptides as they exhibited the above mentioned activities. The bioactive peptides were also characterized and profile of its amino acid content was determined. The presence and characterization of bioactive peptides were confirmed by Tricine-SDS-PAGE and LC-MS-MS analysis. The results were analyzed for the presence of essential amino acids. The results indicated that the protein peptides have potential applications as food supplements. As the protease hydrolyzed peptides containing hydrophobic amino acids, are much useful in reducing the bitterness by removing these amino acids. Therefore, Bacillus sp. protease may have the scope to eliminate the bitter taste by selective hydrolysis of bitter peptides and may offer possibilities for debittering of protein hydrolysates and utilization of non-conventional food sources.